Anke Stuurman, Margarita Riera, Kaatje Bollaerts, Maria Alexandridou, Topi Turunen, Caterina Rizzo, Hanna Nohynek, Ulrike Baum, Ritva Syrjänen, Uy Hoang, Miriam Levi & the DRIVE Consortium.
Introduction
This report presents an executive summary of how the European DRIVE research platform for brand-specific influenza vaccine effectiveness estimates was tested during the 2017/18 influenza season. DRIVE (Development of Robust and Innovative Vaccine Effectiveness) is a public-private partnership that aims to answer the new European regulatory requirements for influenza vaccines by providing brand-specific influenza vaccine effectiveness data annually.
The results of the DRIVE collaboration will serve the regulators, healthcare professionals as well as the society at large. A detailed description of the season and the various pilot vaccine effectiveness analyses conducted by DRIVE is provided in the DRIVE deliverable 7.4: First seasonal final report of conducted studies.
Background
A need for brand-specific influenza vaccine effectiveness estimates
Influenza is a major public health problem. Vaccines are the cornerstone of preventing influenza illness, but their effectiveness can vary from year to year and across recipient groups.
While influenza vaccine effectiveness is being evaluated annually in many EU member states, the results are generally not specific or robust enough to gain a thorough understanding of influenza vaccine effectiveness by type and brand. There is an increasing number of different types of influenza vaccines available, which differ in composition and manufacturing process. The efficacy of these vaccines has been established in clinical studies, which form the basis of their licensure, however, vaccine performance in real word settings can vary between different groups within given populations and from season to season due to a number of factors.
The main aim of DRIVE is not to study differences in IVE between different brands, but rather understand further how individual vaccines perform in the context of annual recommendations. Thus, DRIVE is part of an ongoing effort to monitor the benefit and risk of influenza vaccines from a regulatory perspective.
DRIVE can help demonstrate the value of influenza vaccination, but also to potentially identify vaccines with consistent suboptimal responses for some subgroups of the population or support signal detection. The European Medicines Agency (EMA) has issued a new guideline that came into force in 2017¹. It requests influenza vaccine effectiveness (IVE) evaluation for all influenza vaccine brands used in Europe each season. Responding to these regulatory requirements is the responsibility of the marketing authorization holders. However, brand and particularly type-specific IVE estimates are also of public health importance. Since many European public health institutions have extensive experience of IVE studies, EMA has encouraged the industry and public health institutes to work together to address these regulatory and public health needs.
Hence, a new public-private partnership named DRIVE (Development of Robust and Innovative Vaccine Effectiveness) was launched by the Innovative Medicines Initiative (IMI) in July 2017 to establish a sustainable platform for brand-specific IVE studies in the European Union.
The DRIVE model
In DRIVE, data from several independently operating national or regional study sites located in different parts of Europe are analysed jointly with the aim of obtaining sufficient geographical coverage and sample size for brand-specific IVE estimates (fig. 1).
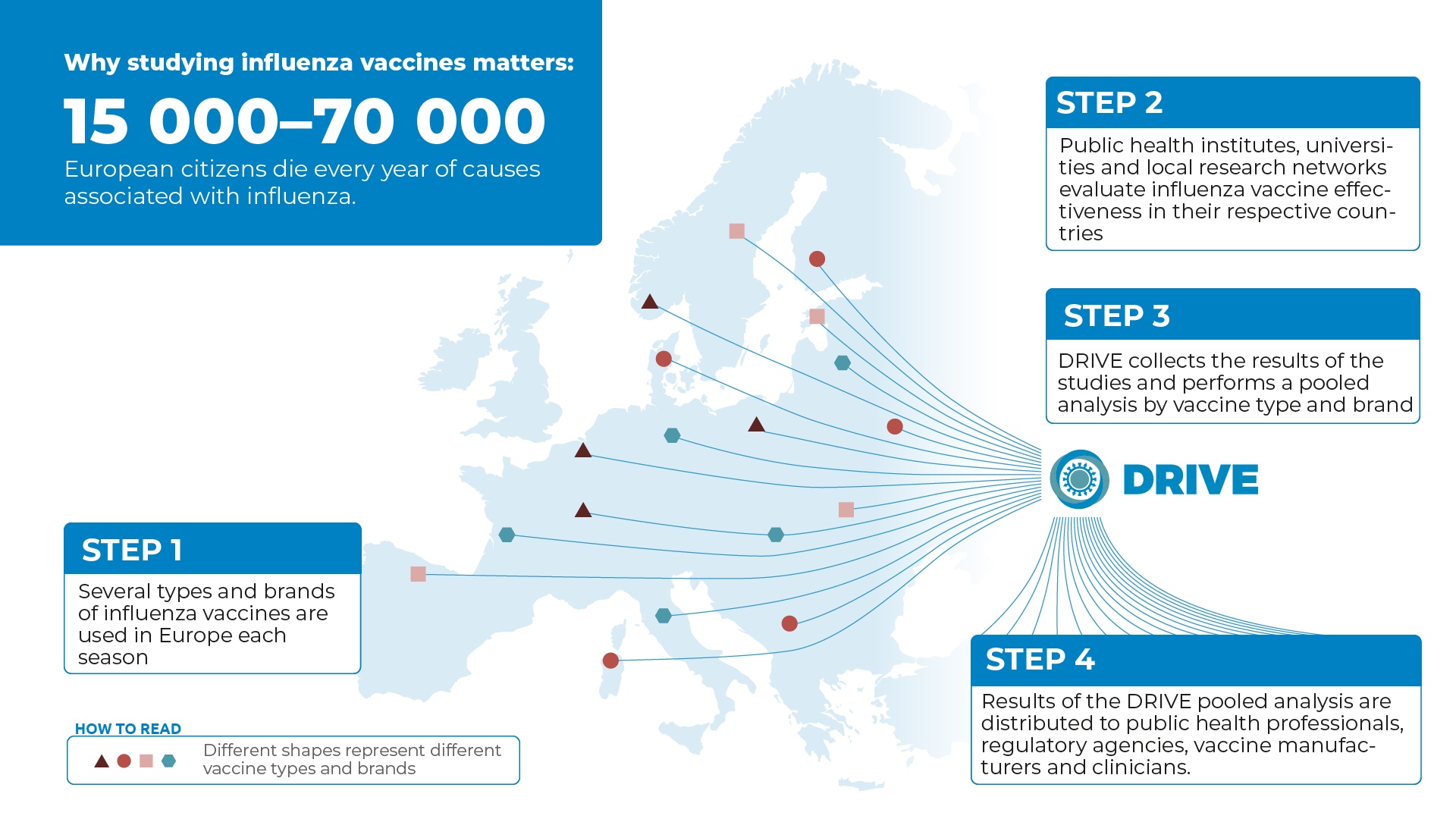
Figure 1. DRIVE collects the results of several European IVE studies and performs a pooled analysis by vaccine brand.
As understanding the effectiveness of influenza vaccines is a priority for both those in charge of public health and vaccine manufacturers, DRIVE operates under a public-private partnership. The studies themselves are conducted independently by public partners, without involvement of vaccine manufacturers. In the interest of scientific independence, rigour and transparency an Independent Scientific Committee (ISC) of external experts oversees the studies and the resulting scientific outputs of DRIVE. Once written by the public partners, the vaccine manufacturers review the scientific outputs through a transparent and traceable written process, however the ISC ultimately
decides upon the inclusion of the vaccine manufacturers’ comments.
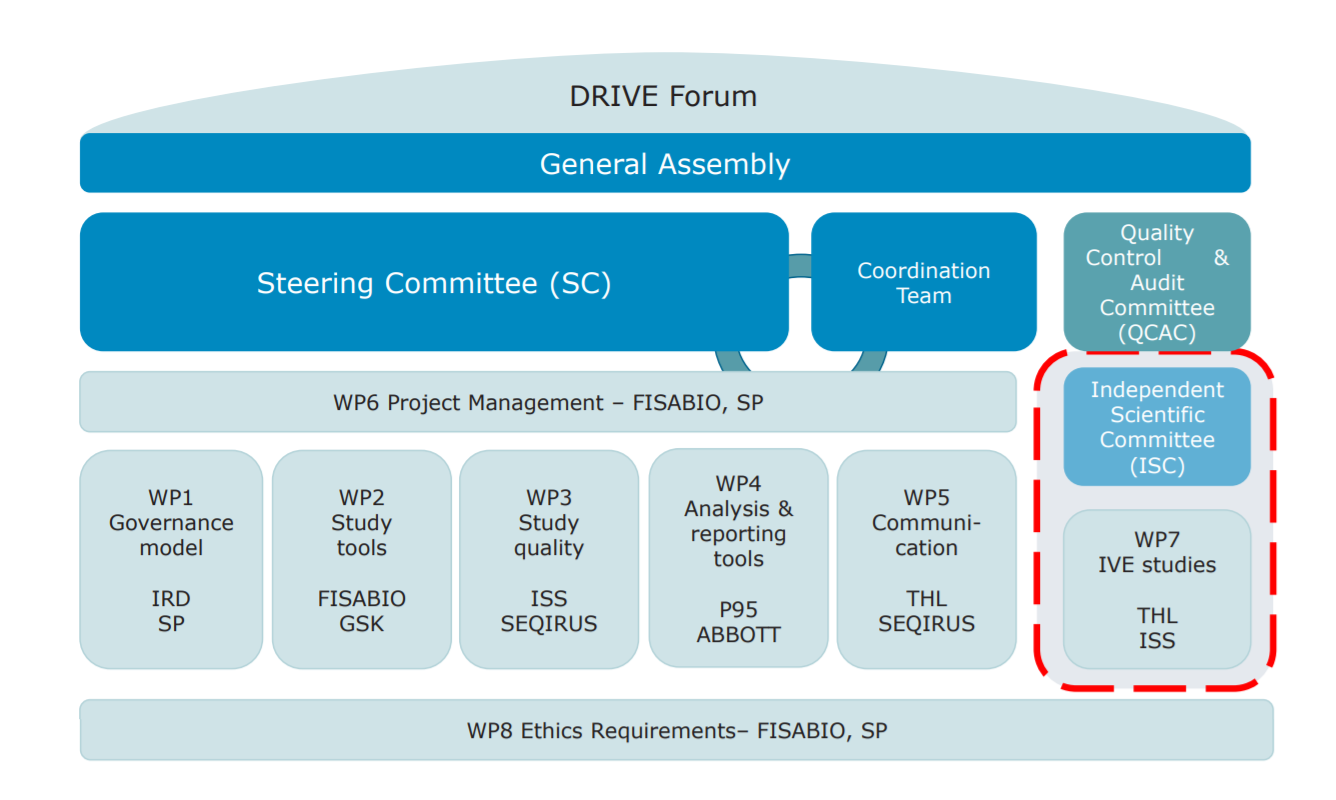
Figure 2. DRIVE is governed by a Steering Committee and ultimately, a General Assembly. Work Package 7, responsible for the studies, operates independently from vaccine manufacturers. The other Work Packages, co-led by public and private partners, perform supporting tasks such as quality assurance, communications and mapping where the different brands are used.
DRIVE governance
DRIVE consortium
Influenza season 2017/18 in Europe
Based on the WHO recommendations on strains to include in seasonal influenza vaccines, the vaccines used in the Northern hemisphere in the 2017/18 season contained the following strains:
- A/Michigan/45/2015 (H1N1)pdm09-like virus
- A/Hong Kong/4801/2014 (H3N2)-like virus
- B/Brisbane/60/2008-like virus (B/Victoria lineage)
- B/Phuket/3073/2013-like virus (B/Yamagata lineage; only in quadrivalent
vaccines).
WHO: Recommended composition of influenza virus vaccines for use in the 2017-2018 northern hemisphere influenza season https://www.who.int/influenza/vaccines/virus/recommendations/2017_18_north/en/.
Based on the surveillance data generated by ECDC with the contributions of the Member States, influenza viruses circulated at high levels between weeks 51/2017 and 13/2018².
The majority were influenza B viruses of the B/Yamagata lineage, which was not included in the trivalent influenza vaccines. Different patterns of dominant virus types and A subtypes were observed between the European countries. Country-specific descriptions of the epidemic are included in pilot year report.
Methods
The first year of DRIVE was dedicated to establishing and testing the IVE research platform through a pilot study. The study was not designed to generate robust IVE estimates but rather to refine the methodology for future studies to ensure DRIVE can meet its intended aim.
Overview of the study sites
Five study sites from four countries (Austria, Finland, Italy & Spain) contributed data to the DRIVE pilot study. Study sites used their own protocols for data collection, as the influenza season started only a few months after the launch of the DRIVE project. Inclusion and exclusion criteria were harmonized where possible at the time of data analysis. Apart from study design, the studies differed with respect to healthcare setting, catchment area, swabbing strategy of influenza-like illness (ILI) cases, ILI case definitions, age groups, laboratory tests performed, and variables available to adjust for confounding. An overview of the most important study site characteristics is given in Figure 3.
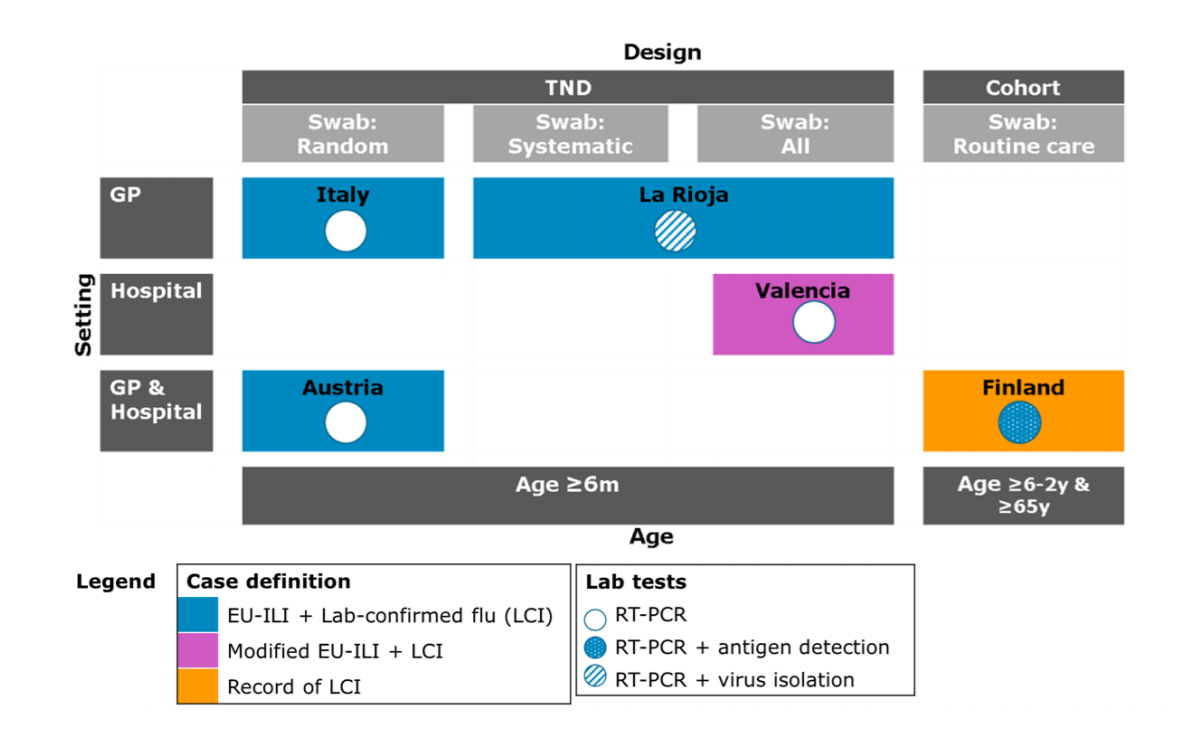
Figure 3. Characteristics of study sites included in the pilot, 2017/2018. In La Rioja, the first 2 ILI patients aged <65 years each week who provided informed consent were swabbed; whereas all ILI patients aged ≥65 years who provided informed consent were swabbed.
Pooled analysis
Influenza VE brand estimates were calculated using a two-stage pooling approach. In a first step, crude and confounder adjusted site-specific IVE estimates were calculated. In a second step, a random effects meta-analysis of odds ratio estimates (TND studies) or log-transformed relative risk estimates (cohort study) of IVE was conducted. IVE was adjusted for confounders including sex, age group, hospitalizations in the previous 12 months (except for Austria), influenza vaccination in the previous season and the presence of at least one chronic condition but only if retained from the model build.
More information on the study design and statistical methods can be found in Pilot year report (DRIVE deliverable D7.4) and its annexes (available at www.drive-eu.org). All analyses were performed centrally at the DRIVE server.
Results
Assembly of the DRIVE research platform
A separate work package (WP7) consisting of non-industry organizations was set up to carry out the studies, whose results are evaluated by an Independent Scientific Committee (ISC). Harmonized study protocols for TND and cohort studies (D7.1 & D7.2) were developed
during the 2017/18 season and will be implemented in the 2018/19 season. A framework for data analysis (D4.1), a data management plan (D4.2), a report template (D4.3) and a generic Statistical Analysis Plan (SAP) (D4.4) were written during the first year of the DRIVE
project. P95 provided the IT infrastructure needed to share, access and analyse data and has built an Electronic Study Support Application (D2.3) which will also be implemented in the 2018-19 season. The ISC was assembled in January 2018.
DRIVE governance
DRIVE deliverables
Influenza vaccine effectiveness estimates
Overall, data were collected on 2,573 cases and 2,426 controls in the TND studies and 241,394 person-years for vaccinated subjects and 288,655 person-years for non-vaccinated subjects in the register-based cohort study. Information on vaccine brand used was successfully collected at all sites except Austria where the brand was unknown for 55% of vaccinated subjects. IVE estimates by vaccine type, vaccine brand and overall as well as by host-related covariates, type of influenza outcomes and study characteristics were calculated in accordance to the SAP.
The results of the DRIVE analyses of IVE by any vaccine, vaccine type and brand (all anonymized due to the pilot nature of the first year) are shown in forest plots and meta-analyses in figures 4–8. IVE by influenza virus type and subtype is shown in figure 9. Due to limited sample size, brand-specific pooled analyses were only performed for two brands. In addition, IVE was estimated for two brands included in the Finnish cohort study.
Results of the DRIVE analysis: Figures 4-8
Discussion
The influenza season of 2017/18 was considered a pilot for the DRIVE consortium to test the feasibility of its research platform for brand-specific IVE studies. A separate work package consisting of non-industry organizations was set up to design and carry out the studies, whose results were evaluated by an Independent Scientific Committee.
The multi-country study included one register-based cohort study and four test-negative design studies. Influenza VE estimates were calculated using a two-stage pooling approach. This pilot study comes with limitations. Due to lack of generic protocols, there were methodological differences between the study sites. Some differences in effectiveness estimates may also be due to variability in study populations or influenza epidemiology.
While a total of 11 brands were covered by the studies, the sample size only allowed IVE estimates for four of them. Because of limited statistical power, limited ability to stratify by age and study setting and differences between studies, the type- and brand-specific estimates are not directly comparable with each other. For these reasons, the results of this pilot year cannot be used to inform medical or regulatory decision-making on any influenza vaccine brand per se.
Conclusion
The DRIVE platform was successfully established during the influenza season 2017/18. Brand information was captured successfully and comprehensively by all but one site. Even with the limitations described above, the results demonstrated the general feasibility of evaluating brand-specific IVE in a multicenter setting.
In the 2018/19 season, the number of study sites included in the platform will more than double, data collection will be harmonized through generic protocols, and analysis will be stratified by study setting and age group. This is intended to overcome some of the main limitations identified during the 2017/2018 pilot seasons and is anticipated to further increase the possibility to generate robust pooled IVE estimates for more brands in the future.
Download the Executive summary as PDF
Read more at DRIVE website
- The full report, appendices and the comments by the Independent Scientific Committee
- The summary for media and the general public
Follow @drive_eu at Twitter and LinkedIn – and join the discussion with the hashtag #DRIVEflu.
The DRIVE project has received funding from the Innovative Medicines Initiative 2 Joint Undertaking under grant agreement No 777363. This Joint Undertaking receives support from the European Union’s Horizon 2020 research and innovation programme and EFPIA.
http://www.imi.europa.eu
1. ECDC Surveillance Report. Annual Epidemiological Report for 2017 Seasonal influenza, 2017–
2018, available at: https://ecdc.europa.eu/sites/portal/files/documents/seasonal-influenza-annual-epidemiological-report-2017.pdf. Accessed 4.12.18.
2. ECDC Surveillance Report. Annual Epidemiological Report for 2017 Seasonal influenza, 2017–
2018, available at: https://ecdc.europa.eu/sites/portal/files/documents/seasonal-influenza-annual-epidemiological-report-2017.pdf. Accessed 4.12.18.
